The two phases exiting the column are collected in the appropriate return tank. The density difference between the phases causes counter-current flow in opposite directions through the packed column where the acid is removed from the organic component into the aqueous component. Demineralised water is pumped from the aqueous feed tank to the top of the extraction column. The 1-Octanol containing up to 1.5% of Propionic acid is pumped from the organic feed tank to the base of the extraction column. The organic circuit uses a variable speed diaphragm pump that is air powered and contains no electrical components allowing safe operation using organic solvents in such as 1-Octanol. Conductivity and temperature sensors in both the feed and return lines allow the amount of acid transferred to the aqueous phase to be determined. The aqueous circuit includes a variable speed rotary diaphragm pump with built in rotation counter that allows the flow rate to be determined. Four process tanks are included, a feed tank and collection tank for the organic phase and a feed tank and collection tank for the aqueous phase. The UOP5-MkII comprises a glass separation column, 1600mm long and 50mm diameter, packed with glass Raschig rings which provide a large surface area for the extraction process. The Armfield UOP5-MkII introduces a system using 1-Octanol or Canola oil which are safe, readily available and inexpensive. 
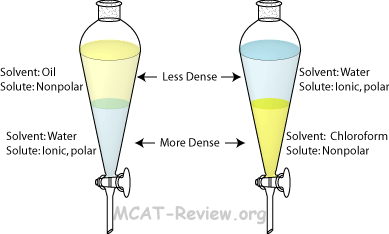
Traditionally this has been difficult to demonstrate in the laboratory, requiring the use of highly toxic, expensive and / or environmentally damaging solvents. It is important to note that the solute will still be present in both phases, and multiple rounds of extraction may be required to transfer a high yield of the solute into the extraction solvent.Liquid / liquid extraction is an important operation in chemical engineering where the separation of one or more of the components from a liquid mixture is required. For example, if an organic compound (such as caffeine) is dissolved in an aqueous solvent (water) and then mixed with an organic solvent (such as dichloromethane) the caffeine will preferentially partition into the organic solvent as it is more soluble in this phase. The solubility of a solute of interest is based upon the principle of ‘like dissolves like’ in that, a highly polar solute will be more soluble in a highly polar solvent, and vice versa. The relative solubility of a solute between two immiscible solvents can be measured as the partition coefficient (P), the ratio of concentrations of a solute between two immiscible solvents at equilibrium. It is important to ensure that the solute is more soluble in the extraction solvent than the other solvent, so it will preferentially partition into this phase. The solute will distribute itself in a certain, predictable, ratio between the two immiscible solvents depending on how soluble it is in each. The method of liquid-liquid extraction is based on the concept of relative solubility. The two immiscible liquid phases are typically water (aqueous phase) and an organic solvent (organic phase) and the extraction is most commonly carried out using a separatory funnel. Here, a solute of interest is extracted from one liquid phase into another immiscible liquid phase, in which it is relatively more soluble. Liquid-liquid extraction is a separation method based on the physical properties of different compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
